Homolytic and Heterolytic Fission
Bond breaking is an important part of organic chemistry –
Bond Fission:
a. Homolytic fission: Each atom separates with one electron, leading to the formation of highly reactive entities called radicals, owing their reactivity to their unpaired electron.
b. Heterolytic fission: One atom holds on to electrons, leaving none for the other, the result in the above case being a negative and positive ion, respectively, the result being the formation of an ion pair. Reactions involving radicals tend to occur in the gas phase and in solution in non-polar solvents and to be catalyzed by light and by the addition of other radicals. Reactions involving ionic intermediates take place more readily in solution in polar solvents, because of the greater ease of separation of charges therein and very often because of the stabilization of the resultant ion pairs through solvation.
Question
Which type of fission is this?
Ans: It is heterolytic fission.
Ans: It is heterolytic fission.
Bond cleavage or scission is the splitting of chemical bonds.
In general, there are two classifications for bond cleavage: homolytic and heterolytic, depending on the nature of the process.
In homolytic cleavage, or homolysis, the two electrons in a cleaved covalent bond are divided equally between the products. This process is also known as homolytic, or radical fission.
In heterolytic cleavage, or heterolysis, the bond breaks in such a fashion that the originally-shared pair of electrons remain with one of the fragments. This process is also known as heterolytic, or ionic fission.
The term "bond dissociation energy" refers to the amount of energy required to cleave a bond.
Homolytic bond cleavage (homolytic cleavage; homolysis): Bond breaking in which the bonding electron pair is split evenly between the products. Homolytic cleavage often produces radicals.
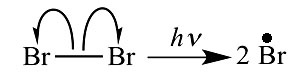
Heterolytic bond cleavage (heterolytic cleavage; heterolysis): Bond breaking in which the bonding electron pair is split unevenly between the products. Heterolytic cleavage often produces at least one ion.
Examples of Heterolytic Bond Cleavage

Heterolysis of a carbon-leaving group bond is the rate-limiting step in the SN1 and E1 mechanisms.
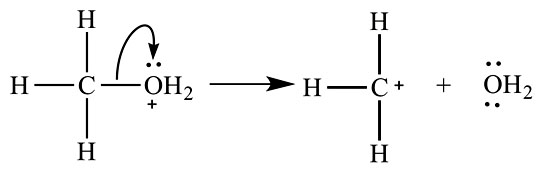
Heterolysis of this oxonium ion produces a carbocation and water.

This mechanism step shows heterolysis of a Lewis acid-base complex. In this case the heterolysis products do not include an ion.
Comments
Post a Comment